Snow
Overview
What is snow?
Snow is precipitation that forms when water vapor freezes. It is not frozen rain, but a crystallization of ice that typically forms in clouds.
When temperatures drop below 0°C (32°F) and there is sufficient humidity in the atmosphere, water vapor condenses directly into ice without going through the liquid stage. Once an ice crystal has formed, it absorbs and freezes additional water vapor from the surrounding air, growing into a snow crystal or snow pellet, which then falls to Earth.
Snow falls in several forms:
- Snowflakes are clusters of ice crystals that fall from a cloud.
- Snow pellets, or graupel, are opaque ice particles in the atmosphere. They form as ice crystals fall through supercooled cloud droplets, which are below freezing but remain a liquid. The cloud droplets then freeze to the crystals, forming a lumpy mass. Graupel tends to be soft and crumbly.
- Sleet is composed of drops of rain or drizzle that freeze into ice as they fall, and is sometimes called a wintery mix of rain and snow. These small, translucent balls of ice are usually smaller than 0.76 centimeters (0.30 inches) in diameter. Official weather observations may list sleet as ice pellets. In some parts of the United States, the term sleet can refer to a mixture of ice pellets and freezing rain.
- One form of precipitation, hail, while frozen, is not considered snow. Hail tends to be larger than sleet, and is usually generated during thunderstorms, which happen more often in spring and summer than in winter. Hailstones form when upward moving air in a thunderstorm, or updrafts, prevent pieces of graupel from falling. Drops of supercooled water hit the graupel and freeze to it, causing the graupel to grow. When the balls of ice become too heavy for the updrafts to continue supporting them, they fall as hailstones.
Because snow is composed of frozen water, or ice, it can also be classified as a mineral. A mineral is a naturally occurring homogeneous solid, inorganically formed, with a definite chemical composition and an ordered atomic arrangement. Ice is naturally occurring, given a temperature below 0 °C (32 °F). It is homogeneous (of one material), formed inorganically, and has an ordered atomic structure. Ice has a definite chemical composition (H20), with hydrogen and oxygen atoms bonding in a specific manner.
Where does it snow?
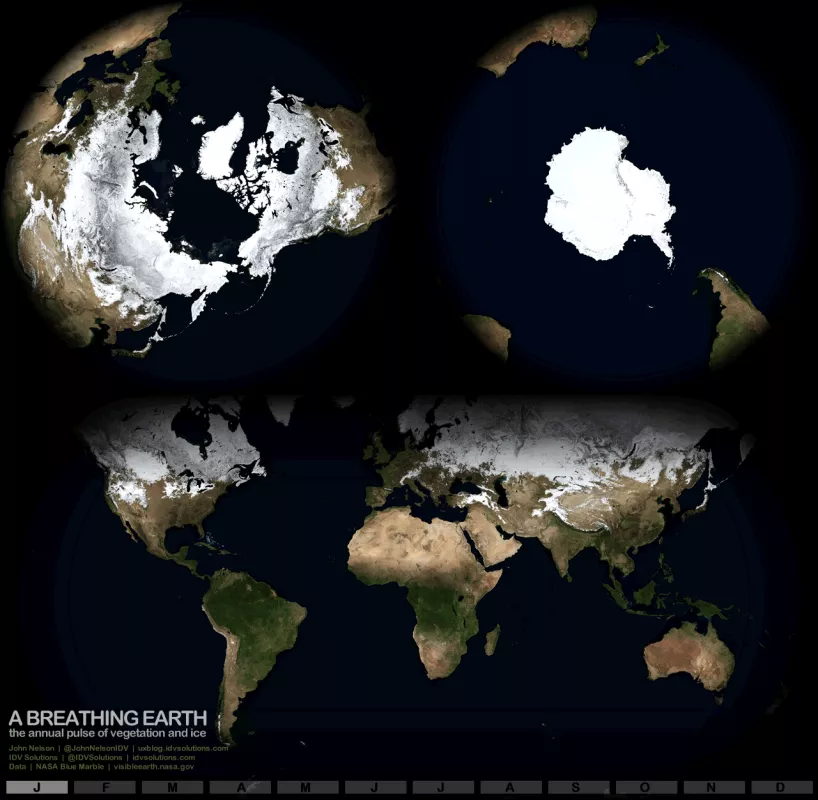
Snow requires two specific weather conditions: low temperatures and moisture in the atmosphere. In warm, humid places, such as Florida, there is significant moisture in the air, but temperatures are rarely low enough to produce snow. And while many deserts get quite cold in the winter, there is rarely enough moisture in the atmosphere to produce snow. Even Antarctica, the coldest and iciest continent, contains a region called the Dry Valleys, where it is extremely cold, but so dry that snow hardly ever falls.
Snow is most common in high altitudes and high latitudes, particularly among the mountainous regions of the Northern and Southern Hemispheres. Annually, snow covers as much as 46 million square kilometers (about 17.8 million square miles), particularly over North America, Greenland, Europe, and Russia. Nearly every location in the United States has seen snowfall. Even portions of Florida have received a few snow flurries.
Snow also falls in the Southern Hemisphere during the austral winter, primarily in Antarctica and in the high mountains of New Zealand and South America.
Local and regional snowfall patterns
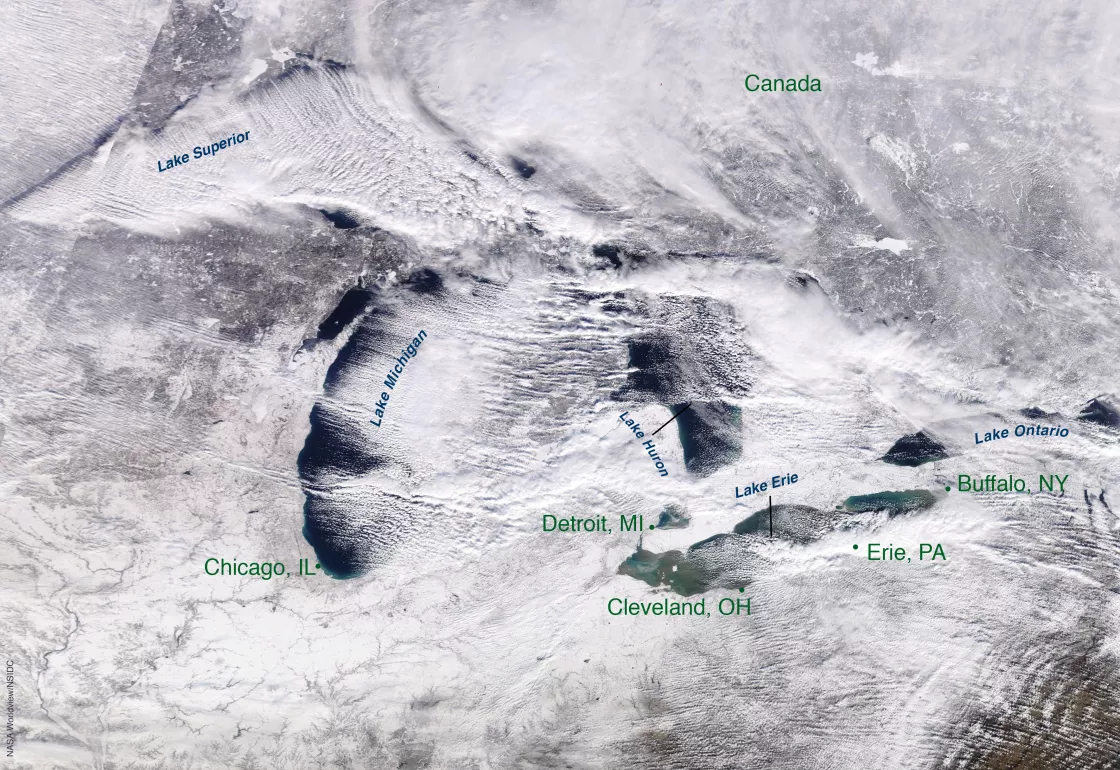
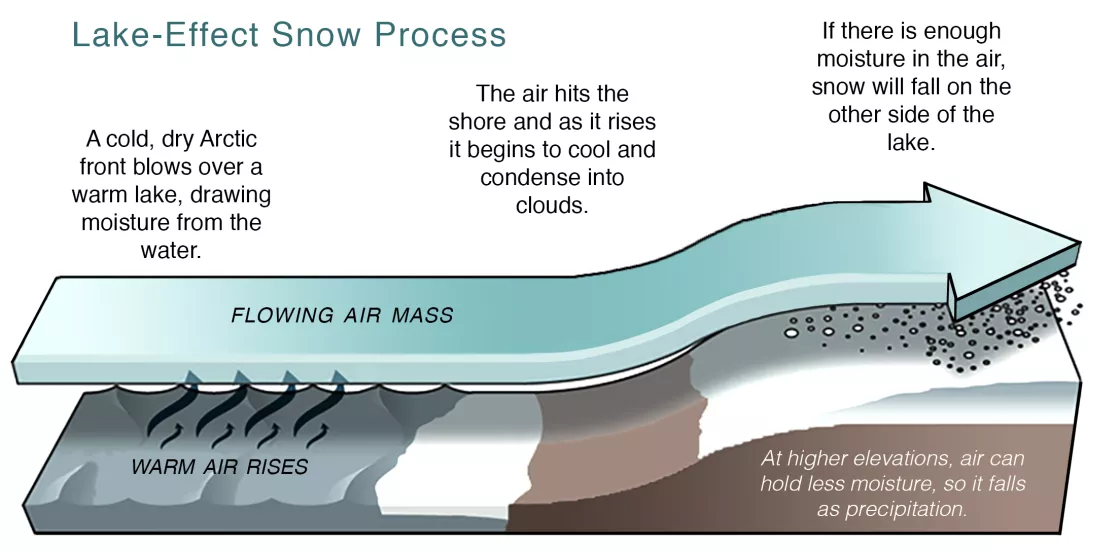
Although snow is commonly associated with mountains, other geographical features can promote the formation of snow. Large lakes, for instance, can produce locally heavy snow, often called lake-effect snow. As cold, dry air passes over the lakes, it picks up moisture and heat from the relatively warm water. The heated air rises, cools off, and the moisture condenses to form clouds. If these clouds contain enough moisture, they will produce snow once they come into contact with land. Lake-effect snow is common along the southern and eastern edges of the Great Lakes in the United States and near the Great Salt Lake in Utah, as well as around some of the large lakes in Canada and Europe. The United Kingdom, Japan, and Korea also experience similar phenomena, but the snow-producing moisture comes from the surrounding oceans instead of lakes.
Winter weather can sometimes create turbulent conditions as the air heats and rises or cools and descends. A rare and strange byproduct of this turbulence is thundersnow, which is a snowstorm with thunder and lightning.
Local weather patterns can also produce snow. For instance, the northeastern coast of the United States is often battered by storms called Nor'easters, which produce heavy snow, rain, and waves that crash onto the beaches.
Terrain and snow
How much snow accumulates in a particular area depends on much more than latitude and time of year. Differences in elevation, vegetation, proximity to a coast, and prevailing wind and weather patterns create different “snow climates.” While Utah and Colorado skiers often enjoy relatively dry, fluffy “champagne powder,” for instance, California skiers usually work with wetter “Sierra cement.”
But differences in terrain and wind cause substantial changes in snow climates over much shorter distances. Wind can scrape snow off a slope and deposit the snow on the other side, causing a tenfold difference in snow depth over the same mountain.